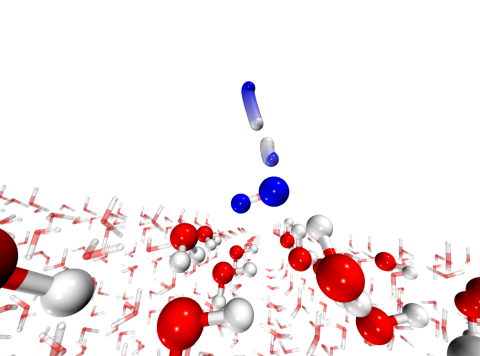
Chemical reactions in the interstellar medium occur at low temperature, often on ice surfaces. Those are found to enhance chemical reactivity. In combination with atom tunnelling, reactions with a significant activation barrier can take place even at the cryogenic temperatures of outer space.
To model the chemical environment of water formation, we calculated rate constants with semiclassical instanton theory and the QM/MM approach. For that, ChemShell was a crucial tool enabling easy setup and communication between the electronic structure calculations, the force field, and the DL-FIND geometry optimisation library.
We found that atom tunnelling is responsible for this reaction to proceed with a rate constant more than three orders of magnitude higher than classically expected at a temperature of 150 K. These reaction rate constants are currently used to model the chemical kinetics in interstellar objects, ranging from molecular clouds to protoplanetary disks.
J. Meisner, T. Lamberts, and J. Kästner, “Atom Tunneling in the Water Formation Reaction H2 + OH → H2O + H on an Ice Surface“, ACS Earth Space Chem., 2017, 1, 399-410.
